About me
I specialize in the intersection of quality engineering and regulatory affairs for medical devices.
A passionate quality champion transforming medical device compliance into inspiring success stories. I bring hands-on expertise in EU MDR transitions and CAPA implementations, breathing life into quality systems through innovative solutions. From redesigning device labels to navigating complex regulatory submissions, I blend technical prowess with genuine care, ensuring patient safety and product excellence go hand in hand.
Specializations
-
Regulatory Affairs
-
Quality Engineering
-
Medical Devices
-
Pharmaceutical Sciences
My Professional Superpowers
-
Labeling
1. Design labels & IFUs to integrate UDI data matrix/CE Marking requirements.
2. Ensure compliance with ISO 15223, ISO 20417, IEC 60601, FDA Standards.
3. Validate regulatory symbols, 2D barcodes, label art proofs.
-
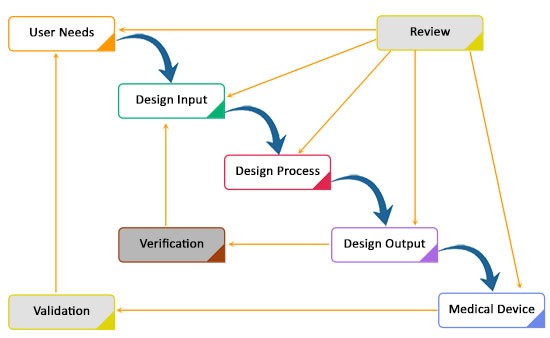
Quality Management System
1. CAPA implementation & root cause analysis techniques (PDCA, Fishbone, 5-Whys)
2. MRB & RMA activities
3. Failure Mode and Effects Analysis
4. ISO 13485 & ISO 9001
5. FDA’s QMSR (21 CFR 820)
6. Risk Management & ISO 14971
7. RoHS, REACH, and CA Prop 65
-
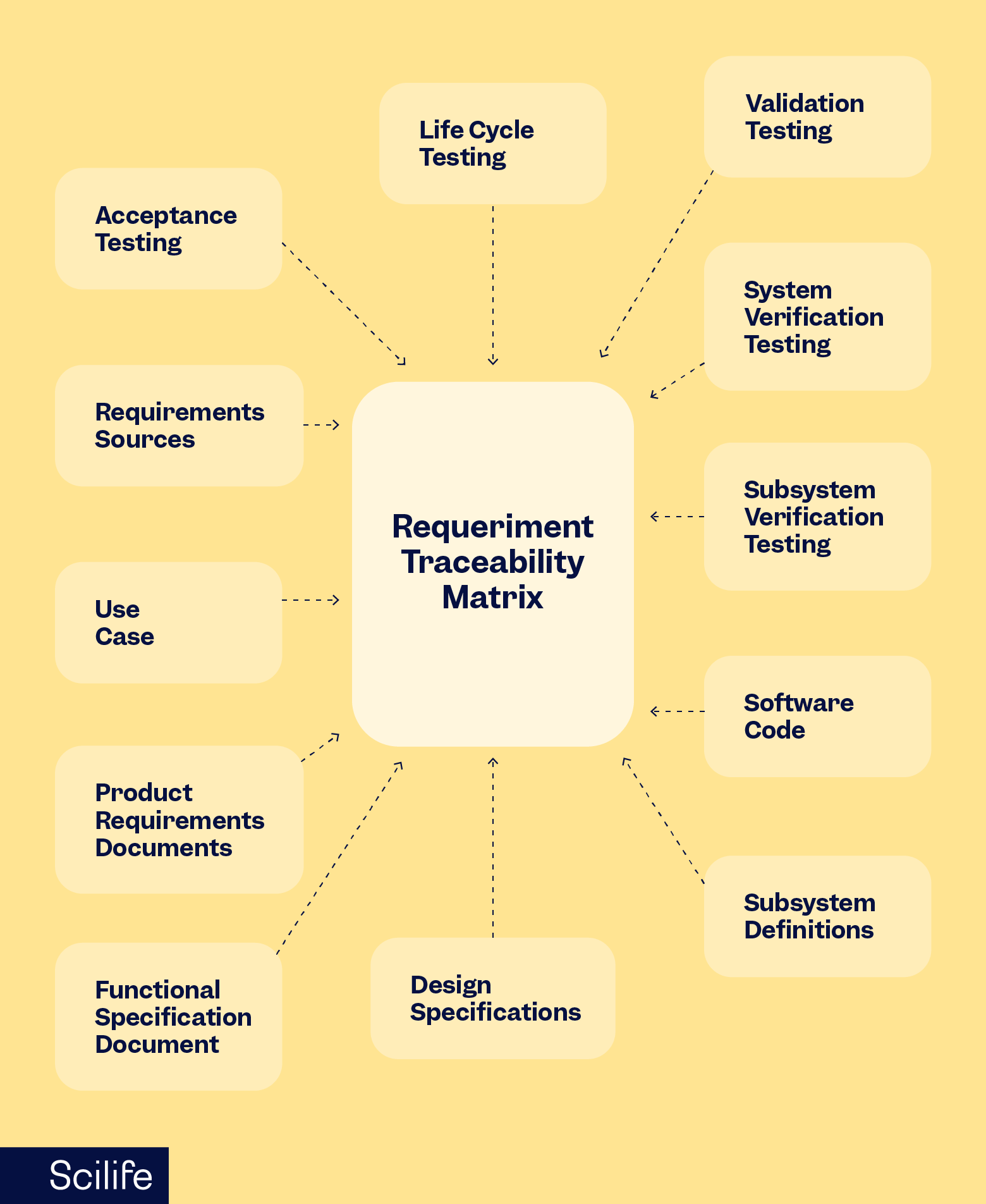
Regulatory Submissions
1. EU MDR Technical Files
2. Biological (BEP, BER), and Clinical (CEP, CER, PMS, PSUR) documentation
3. MAA and CTD preparation
4. FDA 510(k), Letter-to-File & Device Listings
5. EDMS - Veeva Vault
6. EU declaration of conformity
7. Certificate to Foreign Government (CFG)
8. Health Canada Medical Device Licenses
9. Notified Body/CE Marking
10. CDSCO
-
Audits
1. ISO 13485 and INMETRO 384 recertification audits
2. Internal audits
3. Supplier audits
4. MDSAP
Recommendations